WHAT IS MANGANESE?

Manganese is a chemical element with the symbol Mn and atomic number 25. Manganese makes up about 0.1% of the earth’s crust. It is the 4th most used metal on earth in terms of tonnage, behind iron, aluminum and copper. It has numerous applications, including objects made of steel, portable batteries, or aluminium beverage cans. In each case Manganese plays a vital role in improving the properties of the alloys and compounds involved in each specific application. Manganese atomic mass is $54.938044u$.
When Was Manganese Discovered?

Stone Age
The earliest-known human use of Manganese compounds was in the Stone Age when humans used manganese dioxide as a pigment for their cave paintings during the upper paleolithic period, 17.000 years ago.

Ancient Greece
The presence of manganese in the iron ore used by the Spartans perhaps explains why their steel weapons were superior to those of their enemies. Manganese has also long been related to glass-making.

Roman Empire
The Egyptians and the Romans used Manganese ore either ro remove color from glass or to give it pink, purple and black tints. It has been continually used for this purpose until modern times.

17th – 18th Century
The German chemist Glauber obtained permanganate, the first usable manganese salt. Nearly a century later, manganese oxide became the basis for the manufacture of chlorine. Manganese was recognized as an element in 1771, by the Swedish chemist Scheele. The metal was first isolated in 1774 by one of his collaborators, another Swedish chemist, Johan Gottlieb Gahn.

19th Century
Both British and French scientists began considering the use of Manganese in steelmaking, with patents granted in the UK in 1799 and 1808. In 1816, a German researcher observed that Manganese increased the hardness of iron, without reducing its malleability or toughness. In 1826 Prieger in Germany produced a ferromanganese containing 80% Manganese in a crucible.

Mid 19th Century
J.M. Heath produced metallic Manganese in England in about 1840. The following year, Pourcel began industrial-scale production of “spiegeleisen”, a pig iron containing a high percentage of Manganese, and in 1875 he started the commercial production of ferromanganese with a 65% manganese content.

Late 19th Century
Ten years later, in 1866, Sir William Siemens patented the use of ferro-manganese in steelmaking to control the levels of phosphorus and sulphur. Subsequently, and in contrast to all the early work involving Manganese and steelmaking, Leclanché in 1868 developed the dry cell battery containing Manganese dioxide.

20th Century
The history of Manganese has been a stream of new processes and metallurgical/chemical applications developed with a significant impact on markets as diverse as beverage cans, agricultural pesticides and fungicides and electronic circuitry used in consumer products.
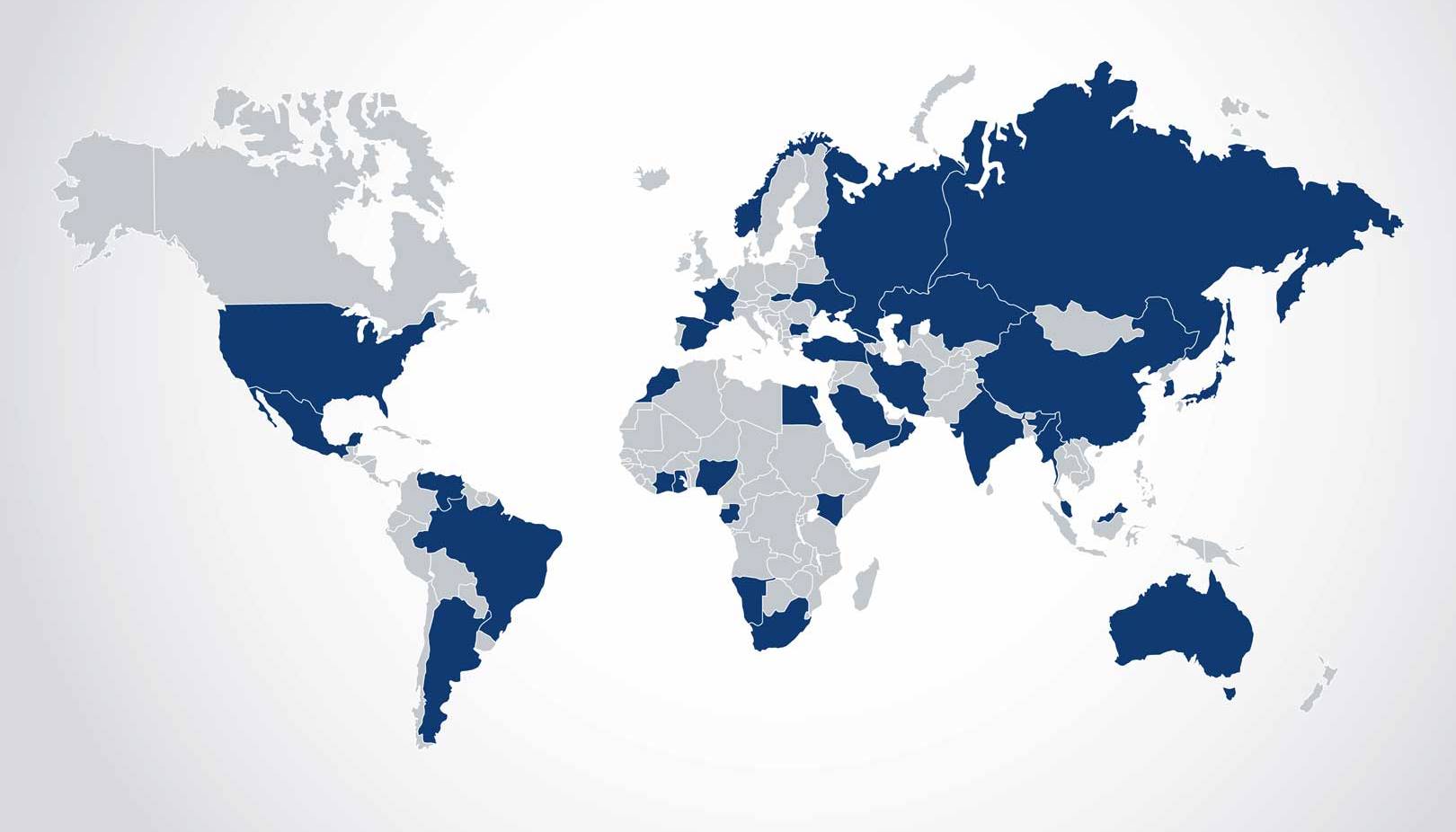
Where Is Manganese Produced?
Major producers of Manganese ore include South Africa, Australia, Brazil, Gabon and Ghana, but Manganese is mined in more than 30 countries, providing jobs for millions of workers, generating billions of dollars in taxes for local countries, and infrastructures for local communities. Both open-pit and underground methods are used to mine Manganese ore, with open-pit methods predominating. Manganese ore is then smelted to make Manganese alloy, with China being by far the biggest producer.
Why Is Manganese Used In Steel?

Manganese is an essential component of steel-making process because it improves certain properties that steel by itself doesn´t have. To name a few, Manganese allows to avoid a phenomenon called “hot shortness”, which causes a surface cracking process at high temperatures, it has an important ability to combine with Sulphur (an unwanted element in steel), and it has a powerful deoxidation capacity. Likewise, Manganese helps to prevent corrosion, make steel more resistant to abrasion, reduces the temperature at which austenite transforms into ferrite, and also increases the hardenability rate.
Why Is Manganese Used In Aluminium?
Aluminum production process is the second most important in terms of Manganese consumption. Small amounts of Manganese are found in aluminum, which enhances corrosion resistance. Aluminum-Manganese alloys and aluminum-manganese-magnesium alloys, have applications in areas such as kitchenware, roofing, car radiators, transportation and most commonly beverage cans (one of the most important markets for aluminum) thanks to the fact that such cans can be recycled.

Why Is Manganese Used In Copper?
In copper alloys production, the use of Manganese result in better mechanical strength as well as improved deoxidize and castability properties. However, copper alloys represent only two million tons per year and hence represents a relative limited market for Manganese demand.
HOW IS MANGANESE USED TO MAKE OTHER METALS?

Manganese is a versatile element, a common component in several alloys’ fabrication. For example, zinc alloys and magnesium alloys contain Manganese, but we can also find this element in gold, silver and bismuth production. Nevertheless, in some cases the quantities involved are very small.
Why Is Mn Used In Batteries?
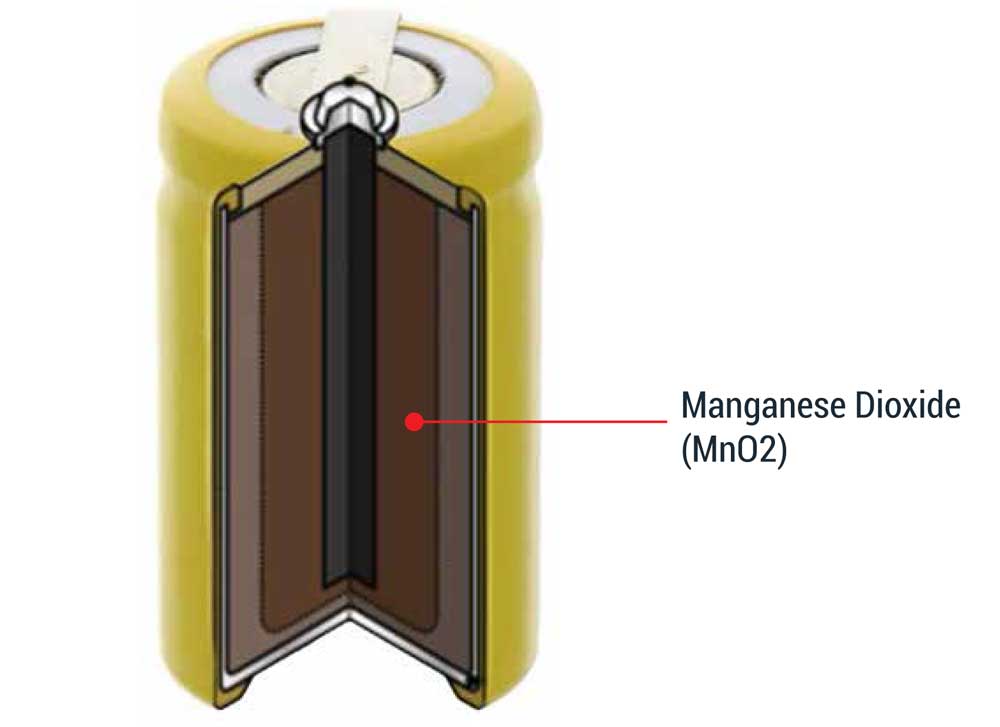
The main non-metallurgical application of Manganese is in the batteries industry. Manganese dioxide is used as a depolarizer in dry-cell batteries fabrication. Worldwide dry cell consumption exceeds 20 billion units per year. The role of the Mn dioxide is to oxidize the hydrogen and form water. Likewise, natural Mn dioxide can also be used in standard cells. The major producing countries of natural Mn dioxide are Gabon, Ghana, Brazil, China, Mexico, and India. These “natural grade battery” ores are ground into a fine powder before being used directly in the cathode mixture.
Why Is Manganese Used In Chemicals?

Manganese is highly used in the chemical industry for several purposes. To name some examples, potassium permanganate is one of the most common Manganese products, it contains oxidant properties ideal for purifying water, waste water treatment and odor control. We also have Mn in the form of “Maneb”, which is sold as a yellow powder for fungicides purposes in agriculture, controlling crop and other diseases. In the food industry, Mn is used as a catalyst in the production of artificial flavors such as vanilla. Manganese sulphate is widely used as an end product in fertilizers and animal feed. Another important material is manganese ferrite, a component widely used in electronics.
What Are Mn Effects On Health?
Naturally ubiquitous in the environment, manganese can be found in rocks, soils, rivers, underground waters and foods such as nuts, legumes, seeds, tea, whole grains, and leafy green vegetables to name but a few. Considered as an essential nutrient, the body requires it to function properly – break down fats, carbohydrates, proteins and for bone strengthening as part of several enzymes.
An essential characteristic of Manganese is that it is a vital element not only for human health, but life overall. It is found mostly in bones, the liver, kidneys and pancreas. Manganese helps the body form connective tissue, bones, blood clotting factors and sex hormones. Several worldwide health organizations recommend daily dietary Manganese intake to ensure a good health. The lack or inadequate levels of Manganese could potentially risk human’s health. For more information about Manganese effect on human health, contact hse@manganese.org
Is There Any Negative Effects To Manganese?
Manganese is found within our own organism, and we need it for various body functions such as absorbing vitamins, producing cartilage in bones and activating certain molecules. Nevertheless, excessive inhalation and/or exposure to high levels of Mn can cause a condition known as manganism. However, thanks to current technologies and labor regulations, as well as measures on safety matters, this is rarely if ever seen today. IMnI ensures that its efforts on these issues are up to date.
100%
SAFE

Is Manganese Safe To Use?

Manganese is an essential trace mineral present in food supply necessary for protein and energy metabolism, bone mineralization, metabolic regulation and cellular protection from oxidative damage. Standard recommendations of Mn are 1.8 mg daily for women and 2.3 mg daily for men aged 19 and older. In particular, women require 2.0 mg of Mn during pregnancy and 2.6 mg during lactation. Consuming Manganese outside these ranges represents a risk to health.


